Anti-Beta-Arrestin [21-B1] Antibody
This mouse IgG1k monoclonal antibody was generated against human ß-arrestin-1 and recognizes human, rat, and hamster beta- arrestin 1 or beta-arrestin 2.
Highlights:
- Recognizes human, rat, and hamster ß-arrestin-1 or ß-arrestin-2 (other species not tested)
- Suitable for Western Blot and Immunoprecipitation applications
Beta-arrestins function in regulating agonist-mediated G-protein coupled receptor (GPCR) signaling by mediating both receptor desensitization and resensitization processes. During homologous desensitization, beta-arrestins bind to the GPRK-phosphorylated receptor and sterically preclude its coupling to the cognate G-protein. Arrestin binding to the receptor blocks further G protein-mediated signaling and targets receptors for internalization, and redirects signaling to alternative G protein-independent pathways, such as ß-arrestin signaling. In addition to GPCRs, arrestins bind to other classes of cell surface receptors and a variety of other signaling proteins.
From the laboratory of Martin Oppermann, MD, University Medical Center Götingen.
This mouse IgG1k monoclonal antibody was generated against human ß-arrestin-1 and recognizes human, rat, and hamster beta- arrestin 1 or beta-arrestin 2.
Highlights:
- Recognizes human, rat, and hamster ß-arrestin-1 or ß-arrestin-2 (other species not tested)
- Suitable for Western Blot and Immunoprecipitation applications
Beta-arrestins function in regulating agonist-mediated G-protein coupled receptor (GPCR) signaling by mediating both receptor desensitization and resensitization processes. During homologous desensitization, beta-arrestins bind to the GPRK-phosphorylated receptor and sterically preclude its coupling to the cognate G-protein. Arrestin binding to the receptor blocks further G protein-mediated signaling and targets receptors for internalization, and redirects signaling to alternative G protein-independent pathways, such as ß-arrestin signaling. In addition to GPCRs, arrestins bind to other classes of cell surface receptors and a variety of other signaling proteins.
From the laboratory of Martin Oppermann, MD, University Medical Center Götingen.
| Product Type: | Antibody |
| Antigen: | Human and rat ß-arrestin-1; ß-arrestin-2 |
| Accession ID: | P49407 |
| Isotype: | IgG1k |
| Clonality: | Monoclonal |
| Clone Name: | 21-B1 |
| Reactivity: | Reacts equally well with (human, rat and hamster) beta- arrestin1 or beta-arrestin2; other species not tested |
| Immunogen: | Human ß-arrestin-1 |
| Species Immunized: | Mouse (Balb/c) |
| Buffer: | 0.1M Sodium Phosphate, pH 7.4, 0.15M NaCl, 0.05% (w/v) Sodium Azide |
| Tested Applications: | WB, IP |
| Storage: | -20C |
| Shipped: | Cold packs |
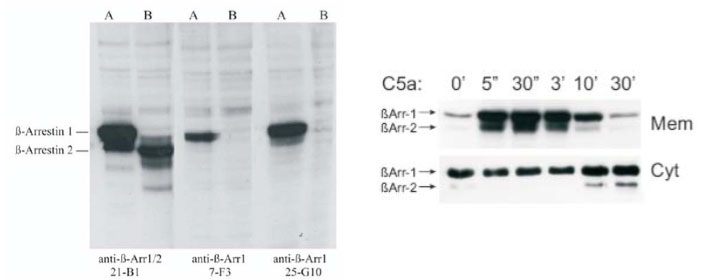
Immunoblots showing the specificity of anti beta-arrestin1/2 mAb 21-B1. Left: Cell lysates from HEK-293 cells overexpressing either rat beta-arrestin1 (A) or rat beta-arrestin2 (B) were probed with 21-B1 and two other mAb specific for beta-arrestin1 (7-F3 and 25-G10). Right: C5a-induced translocation of endogenous beta-arrestin1/2 from the cytosol (Cyt) to the plasma membrane (Mem) in the human myelo-monocytic cell line U937.
- F. Hu?ttenrauch, B. Pollok-Kopp, M. Oppermann. G Protein- coupled receptor kinases promote phosphorylation and beta- arrestin-mediated internalization of CCR5 homo- and hetero- oligomers. J Biol Chem 280:37503 (2005)
- B. Pollok-Kopp, F. Hu?ttenrauch, S. Rethorn, M. Oppermann. Dynamics of protein kinase C -mediated phosphorylation of the complement C5a receptor on serine-334 (Submitted)
If you publish research with this product, please let us know so we can cite your paper.

![Anti-Beta-Arrestin [21-B1] Antibody Anti-Beta-Arrestin [21-B1] Antibody](https://www.kerafast.com/MediaStorage/Product/Images/Medium/1223_2001202001263912220.jpg)
