Recombinant Microbial Glutamate Dehydrogenase (GDH)
Glutamate Dehydrogenase (GDH) produced recombinantly in E. coli.
Highlights:
- Microbial origin - Free from any animal or plant related contamination
- Recombinant source - Fast and sustainable production in a non-pathogenic strain of E. coli
- Extremely reliable- Maintains high activity in a wide range of temperatures (Close to optimal activity between 30C and 70C)
- High thermal stability- Maintains over 85% of its activity for 8 hours at 50C
GDH is an oxidoreductase enzyme which relates carbon and nitrogen metabolism. It catalyzes the reduction of α-ketoglutarate and ammonia to L-glutamate and vice versa. This enzyme is a robust and ideal candidate for research use, and industrial applications in the diagnostics and food industries.
Glutamate Dehydrogenase (GDH) produced recombinantly in E. coli.
Highlights:
- Microbial origin - Free from any animal or plant related contamination
- Recombinant source - Fast and sustainable production in a non-pathogenic strain of E. coli
- Extremely reliable- Maintains high activity in a wide range of temperatures (Close to optimal activity between 30C and 70C)
- High thermal stability- Maintains over 85% of its activity for 8 hours at 50C
GDH is an oxidoreductase enzyme which relates carbon and nitrogen metabolism. It catalyzes the reduction of α-ketoglutarate and ammonia to L-glutamate and vice versa. This enzyme is a robust and ideal candidate for research use, and industrial applications in the diagnostics and food industries.
| Catalog Number | Product | DataSheet | Size | AVAILABILITY | Price | Qty |
|---|
Please contact us for more information about bulk quantities of this product.
| Product Type: | Protein |
| Name: | Glutamate dehydrogenase (GDH) |
| CAS number: | 9001-46-1 |
| EC Number: | 1.4.1.2 |
| Source: | E. coli(recombinant enzyme from a thermophilic bacterium) |
| Molecular Weight: | 270 kDa; Homohexameric ( 45 kDa per subunit) |
| Format: | Lyophilized powder |
| Buffer: | 0.05 M Tris base and 0.5 M NaCl (before lyophilizing) |
| Stability: | At -20ºC, it keeps 100% of its activity after one year |
| Concentration: | > 13% (w/w) |
| Activity: | > 90 U/mg protein; One unit is defined as the conversion of 1?mol of ?-ketoglutarate into glutamate, in 1 minute at 50°C at pH 8.0 |
| Temperature Range: | 20-70ºC (optimal: 50ºC) |
| pH Range: | 7-8.5 (optimal: 8) |
| Storage: | At -20 ºC |
| Shipped: | Cold packs |
Research and Diagnostics:
- Determining levels of urea and ammonia in body fluids (e.g. plasma)
- Control and standard in testing/assay validation
- Positive control in glutamate dehydrogenase detection kits (e.g., for diagnosis of hospital-born infection by Clostridium difficile)
Industry:
- Aroma and flavor development in cheese
- pre-alcoholic fermentation analysis in wine production (e.g., ammonia determination)
- detecting spoilage in meat, fish, seafood, cheese, juices and milk products
- As part of the synthesis process of L-amino acids (e.g. L-glutamine)
Glutamate dehydrogenase activity at different temperatures
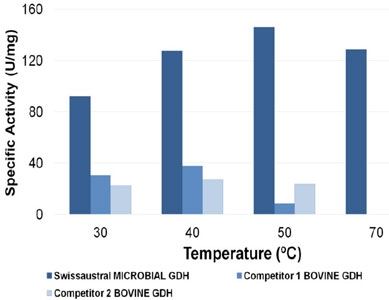
Swissaustral GDH is more stable than GDH from bovine liver sources in a broad spetrum of temperatures offering close tooptimal activity between 30C and 70C.
- D P. Hornby, M J. Aitchison, P C.Engel. (1984).The kinetic mechanism of ox liver glutamate dehydrogenase in the presence of the allosteric effector ADP. The oxidative deamination of L-glutamate. Biochemical Journal 1984-10-01.
- M. Amenábar, J. Blamey. (2011). Purification and characterization of a thermostable glutamate dehydrogenase from a thermophilic bacterium isolated from a sterilization drying oven. Biochemistry and Molecular Biology Reports. 2, 91-95.
- J. DiRuggiero, F. Robb, R. Jagus, H. Klump, K. Borges, M. Kessel, X. Mai, M. Adams. (1993). Characterization, cloning, and in Vitro expression of the extremely thermostable glutamate dehydrogenase from the hyperthermophilic archaeon, ES4. Journal of Biological Chemistry. 268,17767-17774.
- Wong SJ, Ringel AE, Yuan W, Paulo JA, Yoon H, Currie MA, Haigis MC. Development of a colorimetric α-ketoglutarate detection assay for prolyl hydroxylase domain (PHD) proteins. J Biol Chem. 2021 Feb 8:100397. View article
If you publish research with this product, please let us know so we can cite your paper.


