Anti-ADAM15 (mouse), Cytoplasmic Domain Antibody
This rabbit IgG polyclonal antibody was generated against the GST-ADAM15-cytoplasmic tail and is specific for the cytoplasmic domain of mouse ADAM15.
The ADAM (A Disintegrin And Metalloprotease Domain) family is a unique group of proteins that has the ability to cleave or shed extracellular portions of transmembrane proteins. In addition, ADAMs are involved in cell-cell and cell-matrix interactions (e.g., fertilization, muscle development and neurogenesis).
ADAM15 (Metargidin) is a membrane-anchored metalloproteinase that has been implicated in pathological neovascularization and prostate cancer metastasis.
From the laboratory of Carl P. Blobel, MD, PhD, Hospital for Special Surgery.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
This rabbit IgG polyclonal antibody was generated against the GST-ADAM15-cytoplasmic tail and is specific for the cytoplasmic domain of mouse ADAM15.
The ADAM (A Disintegrin And Metalloprotease Domain) family is a unique group of proteins that has the ability to cleave or shed extracellular portions of transmembrane proteins. In addition, ADAMs are involved in cell-cell and cell-matrix interactions (e.g., fertilization, muscle development and neurogenesis).
ADAM15 (Metargidin) is a membrane-anchored metalloproteinase that has been implicated in pathological neovascularization and prostate cancer metastasis.
From the laboratory of Carl P. Blobel, MD, PhD, Hospital for Special Surgery.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
| Product Type: | Antibody |
| Antigen: | GST-ADAM15 cytoplasmic domain |
| Accession ID: | Q13444 |
| Molecular Weight: | ~100 kDa |
| Isotype: | IgG |
| Clonality: | Polyclonal |
| Reactivity: | Mouse |
| Immunogen: | GST-cyto corresponding to the cytoplasmic domain of mouse ADAM15 |
| Species Immunized: | Rabbit |
| Epitope: | Cytoplasmic domain |
| Purification Method: | Serum, not purified |
| Tested Applications: | WB (1:500) |
| Storage: | -80C |
| Shipped: | Dry ice |
| Antibody | Species Reactivity | Specificity |
| ADAM9 | Human | Cytoplasmic Domain |
| ADAM9 | Mouse | Cytoplasmic Domain |
| ADAM12 | Mouse | Cytoplasmic Domain |
| ADAM15 | Human | Cytoplasmic Domain |
| ADAM15 | Mouse | Cytoplasmic Domain |
| ADAM15 | Human | Extracellular Domain |
| ADAM15 | Mouse | Disintegrin Domain |
| ADAM17 | Mouse | Cytoplasmic Domain |
| ADAM17 | Mouse | Pro domain |
| ADAM19 | Mouse | Cytoplasmic Domain |
| ADAM28 | Mouse | Cytoplasmic Domain |
| ADAM28 | Mouse | Extracellular Domain |
| SNX9 | Mouse | Cytoplasmic Domain |
Western Blot Analysis
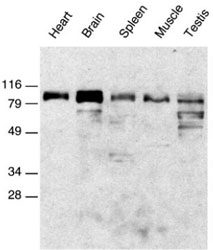
Western blot of reduced glycoproteins from the indicated tissues were probed with ADAM15 cytoplasmic domain antibody.
Adapted from: Lum L, et al. J. Biol Chem. 1998, 273, 26236-26247.
- Lum L.; Reid, M. S.; Blobel, C. P. Intracellular maturation of the mouse metalloprotease disintegrin MDC15. J. Biol Chem. 1998, 273, 26236-26247.
- Horiuchi K, Weskamp G, Lum L, Hammes HP, Cai H, Brodie TA, Ludwig T, Chiusaroli R, Baron R, Preissner KT, Manova K, Blobel CP. Potential role for ADAM15 in pathological neovascularization in mice. Mol Cell Biol. 2003 Aug;23(16):5614-24.
- Maretzky T, Yang G, Ouerfelli O, Overall CM, Worpenberg S, Hassiepen U, Eder J, Blobel CP. Characterization of the catalytic activity of the membrane-anchored metalloproteinase ADAM15 in cell-based assays. Biochem J. 2009 Apr 28;420(1):105-13.
- Maretzky T, Le Gall SM, Worpenberg-Pietruk S, Eder J, Overall CM, Huang XY, Poghosyan Z, Edwards DR, Blobel CP. Src stimulates fibroblast growth factor receptor-2 shedding by an ADAM15 splice variant linked to breast cancer. Cancer Res. 2009 Jun 1;69(11):4573-6.
If you publish research with this product, please let us know so we can cite your paper.