Anti-Complement C3 [8E11] Antibody
This mouse IgG1 antibody was generated against human C3b and iC3b bound to a carbohydrate matrix via the alternative pathway of complement activation and recognizes human C3, C3b and iC3b and cross reacts with primate but not non-primate (guinea pig, horse, dog) homologs.
The complement system consists of a number of small proteins found in the blood, generally synthesized by the liver, and normally circulating as inactive precursors (pro-proteins). When stimulated by one of several triggers, proteases in the system cleave specific proteins to release cytokines and initiate an amplifying cascade of further cleavages. The end-result of this activation cascade is massive amplification of the response and activation of the cell-killing membrane attack complex.
Complement component 3 (C3) plays a central role in the activation of the complement system. C3 cleavage by C3-convertase produces C3b and iC3b. C3b leads to an inflamatory response and also covalently binds to microbial cell surfaces aiding in opsonization of the microbe.
From the laboratory of Ronald P. Taylor, PhD, University of Virginia.
This mouse IgG1 antibody was generated against human C3b and iC3b bound to a carbohydrate matrix via the alternative pathway of complement activation and recognizes human C3, C3b and iC3b and cross reacts with primate but not non-primate (guinea pig, horse, dog) homologs.
The complement system consists of a number of small proteins found in the blood, generally synthesized by the liver, and normally circulating as inactive precursors (pro-proteins). When stimulated by one of several triggers, proteases in the system cleave specific proteins to release cytokines and initiate an amplifying cascade of further cleavages. The end-result of this activation cascade is massive amplification of the response and activation of the cell-killing membrane attack complex.
Complement component 3 (C3) plays a central role in the activation of the complement system. C3 cleavage by C3-convertase produces C3b and iC3b. C3b leads to an inflamatory response and also covalently binds to microbial cell surfaces aiding in opsonization of the microbe.
From the laboratory of Ronald P. Taylor, PhD, University of Virginia.
| Product Type: | Antibody |
| Name: | Anti-Human, Primate Complement C3b, (8E11) Monoclonal Antibody |
| Accession ID: | P01024 |
| Host: | mouse |
| Isotype: | IgG1 |
| Clonality: | Monoclonal |
| Clone Name: | 8 E11 |
| Specificity: | Human C3, C3b and iC3b and cross reacts with primate but not non-primate (guinea pig, horse, dog) homologs |
| Immunogen: | generated against human C3b and iC3b bound to a carbohydrate matrix via the alternative pathway of complement activation |
| Format: | Liquid |
| Purification Method: | Protein G affinity purified |
| Buffer: | PBS, 0.05% Sodium Azide (w/v) |
| Tested Applications: | ELISA |
| Concentration: | 1mg/mL |
| Amount: | 100uL |
| Storage: | Store at 4C |
| Shipped: | Cold packs |
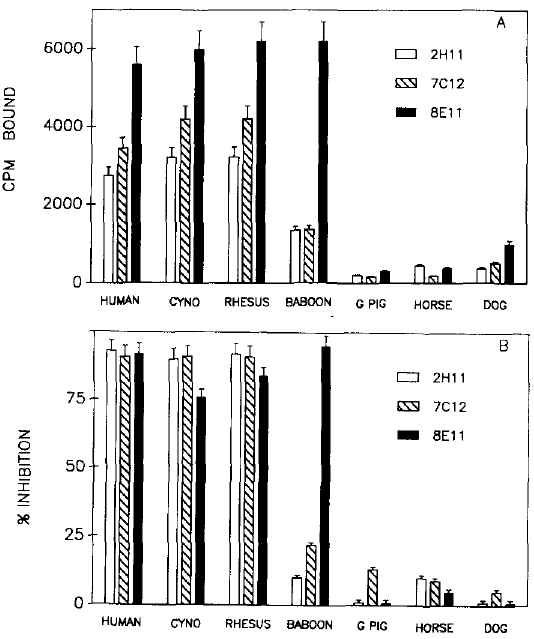
A: Binding of three different nSI-labelled MAbs to C3b(i)-Sepharose prepared with human serum, and to putative C3b(i)-Sepharose samples prepared with the sera of other primates (cynomologous monkey, rhesus monkey, and baboon) and non-primates (guinea pig, horse, and dog). The total cpm input was circa 40000 for each MAb. Other non-primate samples tested, which also showed a lack of binding, were cat, sheep, and cow. B: inhibition of binding of the same 125I-labelled MAbs to human C3b(i)-Sepharose, by zymosan-treated sera (Mollnes and Lachmarm, 1987) of the same species tested in A. MAb 8Ell consistently showed a unique specificity since it was the only MAb of the three that bound baboon C3b(i)-Sepharose (A), and was inhibited by zymosan-treated baboon serum.
Adapted from: Tosic L, Sutherland WM, Kured J, Edberg JC, Taylor RP. Preperation of amonoclona antibodies to C3b by immunization with C3b(i)-sepharose. J Immunol Methods, 120(2):241-249, (1989).
- Tosic L, Sutherland WM, Kured J, Edberg JC, Taylor RP. Preperation of amonoclona antibodies to C3b by immunization with C3b(i)-sepharose. J Immunol Methods, 120(2):241-249, (1989).
If you publish research with this product, please let us know so we can cite your paper.

![Anti-Complement C3 [8E11] Antibody Anti-GFP (Chicken-Standard) Antibody](https://www.kerafast.com/MediaStorage/Product/Images/Medium/113_200120200125541120D.jpg)
