Branched Chain Amino-acid Transaminase-2 (BCAT2); Full-length Human
Branched chain aminotransferase (BCAT2; EC 2.6.1.42) is an aminotransferase enzyme which acts upon branched-chain amino acids. It uses largely α-ketoglutarate in forming branched chain α-keto acids and glutamate. The biological function of branched chain aminotransferases is to catalyse the synthesis of the branched chain amino acids leucine, isoleucine, and valine.
Bulk pricing is available for large quantities upon request. Ask us for a quote.
Branched chain aminotransferase (BCAT2; EC 2.6.1.42) is an aminotransferase enzyme which acts upon branched-chain amino acids. It uses largely α-ketoglutarate in forming branched chain α-keto acids and glutamate. The biological function of branched chain aminotransferases is to catalyse the synthesis of the branched chain amino acids leucine, isoleucine, and valine.
Bulk pricing is available for large quantities upon request. Ask us for a quote.
| Product Type: | Protein |
| Accession ID: | O15382 |
| Source: | E. Coli |
| Molecular Weight: | 44.03 kDa |
| Format: | Liquid |
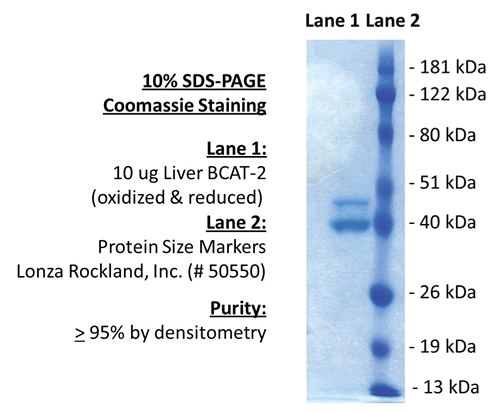
| Purity: | 95% (densitometry) |
| Buffer: | 50 mM potassium phosphate pH-7.4, 50 mM sodium chloride, 0.5 mM ethylenediaminetetraaceticacid, and 2.5% glycerol. |
| Concentration: | 1.0mg/mL |
| Comments: | Human branch-chained aminotransferase-2 , (Accession No. BC004243), aa 390 (full length minus the first 16 aa) with N-terminal hexahistidine tag, MW= 44.01 kDa, expressed in E.coli. |
| Storage: | Stable > 6 months at -80C |
| Shipped: | Dry ice |
SDS-Page:
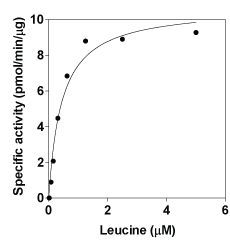
Specific Activity: 10.7 pmol/min/ug. One unit is defined as the amount of enzyme that will convert 1 nmol of NADP to NADPH at 30C. Assay conditions: 100 mM sodium phosphate, pH 7.4, 5 mM DTT, 50mM ammonium sulfate, 5 µM pyroxidal 5 phosphate, 0.5 mM 3-acetyl pyridine adenine dinucleotide, 0.5 mM a-ketoglutarate, 5 mM leucine, 12.5 mM EDTA, 10 units/ml leucine dehydrogenase, and 10 nM human branch-chained aminotransferase-2 at 30C for 30 min.
Enzyme activation: Maximal activity of human branch-chained aminotransferase-2 requires pretreatment of enzyme with 100 mM final DTT for at least 1 hour at 4oC.
If you publish research with this product, please let us know so we can cite your paper.