Anti-Lamin B Receptor (LBR) [BBmLBR 12.F8] Antibody
This mouse IgG2b kappa monoclonal antibody recognizes human and mouse Lamin B Receptor (LBR).
Highlights:
- Reacts with human and mouse Lamin B Receptor (LBR)
- Epitope - Within amino acids residues 1-211 of mouse LBR
- Suitable for Western Blot, Immunohistochemistry, Immunocytochemsitry, and Immunofluorescence applications
Lamin B Receptor (LBR) is a polytopic inner nuclear membrane protein. Its nucleoplasmic domain
interacts with components of the nuclear lamina as well as with chromatin, via HP1. LBR also
exhibits C14 sterol reductase activity. Deficiency of LBR in humans results in Pelget-Huët anomaly
featuring hypolobulated granulocyte nuclei. Homozygous loss of LBR causes Greenberg skeletal
dysplasia a fetal lethal disorder associated with defective cholesterol metabolism.
From the laboratory of Brian Burke, PhD, A*STAR Institute of Medical Biology (IMB).
This mouse IgG2b kappa monoclonal antibody recognizes human and mouse Lamin B Receptor (LBR).
Highlights:
- Reacts with human and mouse Lamin B Receptor (LBR)
- Epitope - Within amino acids residues 1-211 of mouse LBR
- Suitable for Western Blot, Immunohistochemistry, Immunocytochemsitry, and Immunofluorescence applications
Lamin B Receptor (LBR) is a polytopic inner nuclear membrane protein. Its nucleoplasmic domain interacts with components of the nuclear lamina as well as with chromatin, via HP1. LBR also exhibits C14 sterol reductase activity. Deficiency of LBR in humans results in Pelget-Huët anomaly featuring hypolobulated granulocyte nuclei. Homozygous loss of LBR causes Greenberg skeletal dysplasia a fetal lethal disorder associated with defective cholesterol metabolism.
From the laboratory of Brian Burke, PhD, A*STAR Institute of Medical Biology (IMB).
| Product Type: | Antibody |
| Antigen: | Lamin B Receptor |
| Accession ID: | Q14739 |
| Molecular Weight: | Human LBR 70.7 kDa, mouse LBR 71.4 kDa |
| Isotype: | IgG2b kappa |
| Clonality: | Monoclonal |
| Clone Name: | BBmLBR 12.F8 |
| Reactivity: | Human and Mouse, not tested with others |
| Species Immunized: | Mouse |
| Epitope: | Within amino acids residues 1-211 of mouse LBR |
| Buffer: | Cell Culture Supernatant |
| Tested Applications: | WB, IHC, ICC, IF |
| Positive Control: | Hela, U2OS, C2C12, NIE-115 |
| Comments: | Knockout validated |
| Storage: | -80C |
| Shipped: | Cold Packs |
Western blot analysis of BBmLBR 12.F8 antibody.
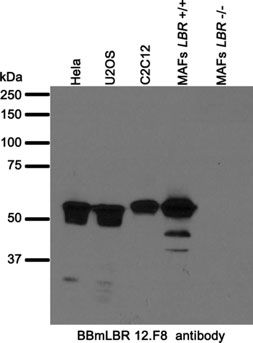
Whole cell lysates from human (Hela, U2OS) and mouse (C2C12, mouse adult fibroblasts (MAFs (7))) cell lines were separated on SDS-PAGE gel, transferred to nitrocellulose membrane and blotted with BBmLBR 12.F8 antibody. Both primary and secondary antibodies were diluted in blocking buffer (5% nonfat dry milk resuspended in 0.1% Tween and 1X PBS). The predicted molecular weight is 70.7 kDa and 71.4 kDa for human and mouse LBR respectively. The absence of a band in LBR-null MAFs indicated that the antibody specifically recognizes LBR. The lower molecular bands detected by the antibody might represent alternatively spliced LBR variants and/or degradation products.
Immunofluorescence staining of small intestine from adult C57BL/6 mice.
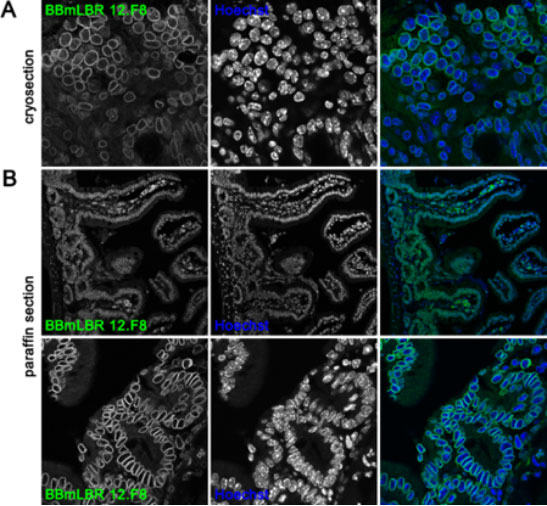
(A) Small intestine was transferred to OCT compound and frozen in liquid nitrogen. Tissue cryosections were fixed in 10% neutral buffered formalin, incubated with 1% Triton X-100 (in PBS) and stained with anti-LBR BBmLBR 12.F8 antibody. (B) Tissue was fixed in 10% NBF and embedded in paraffin. Following heat-induced antigen retrieval (sodium citrate buffer pH 6) the paraffin sections were incubated with 1% Triton X-100 (in PBS) and subsequently stained with mouse monoclonal anti-LBR BBmLBR 12.F8 antibody. Nuclei were visualized with Hoechst. Top and bottom panels in (B) show different magnifications of the small intestine.
Confocal images of U2OS cell fixed with cold methanol (MeOH) (upper panel) or 4% PFA (lower panel) and stained with mouse monoclonal BBmLBR 12.F8 antibody.
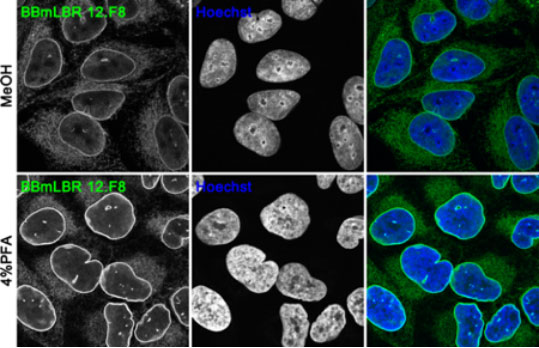
Nuclei were stained with Hoechst. BBmLBR 12.F8 antibody works on cells fixed with 4% PFA and cold MeOH with PFA fixation resulting in a brighter signal of the antibody.
The antibody specifically detects LBR.
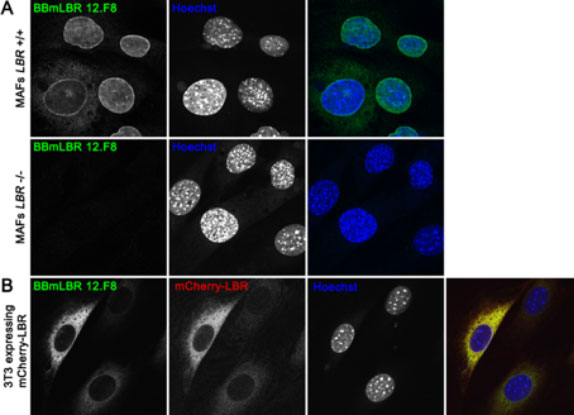
A) Immortalized wild-type mouse adult fibroblast (MAFs LBR+/+) (upper panel) and LBR-null MAFs (MAFs LBR-/-) (7) (lower panel) were fixed in 4% PFA and immunofluorescently stained with BBmLBR 12.F8 mouse monoclonal antibody. In LBR-null MAFs there is a loss of BBmLBR 12.F8 antibody staining signal indicating antibody specificity for LBR. (B) 3T3 cells stably over-expressing mCherry-tagged mouse LBR were fixed in 4% PFA and stained with BBmLBR 12.F8 antibody. The expression levels of mCherry-LBR correspond to intensity of BBmLBR 12.F8 antibody staining. Nuclei were stained with Hoechst.
Immunocytochemistry/Immunofluorescence: formaldehyde, methanol fixation with formaldehyde fixation resulting in a brighter signal
- Worman, H.J., J. Yuan, G. Blobel, S. D. Georgatos. 1988. A lamin B receptor in the nuclear envelope. Proc Natl Acad Sci U S A. 85(22):853-4.
- Ye, Q., H. J. Worman. 1994. Primary structure analysis and lamin B and DNA binding of human LBR, an integral protein of the nuclear envelope inner membrane. J Biol Chem. 269(15):11306-11.
- Ye, Q., H. J Worman. 1996. Interaction between an integral protein of the nuclear envelope inner membrane and human chromodomain proteins homologous to drosophila hp1. J Biol Chem. 271:1465314656.
- Tsai, P.L., C. Zhao, E. Turner, C. Schlieker. 2016. The Lamin B receptor is essential for cholesterol synthesis and perturbed by disease-causing mutations. Elife. Jun 23;5. pii: e16011. doi: 10.7554/eLife.16011.
- Hoffmann, K., C. K. Dreger, A. L. Olins, D. E. Olins, L. D. Shultz, B. Lucke, H. Karl, R. Kaps, D. Müller, A. Vayá, J. Aznar, R. E. Ware, N. Sotelo Cruz, T. H. Lindner, H. Herrmann, A. Reis, K. Sperling. 2002. Mutations in the gene encoding the lamin B receptor produce an altered nuclear morphology in granulocytes (Pelger-Huët anomaly). Nat Genet. 31(4):410-4.
- Waterham, H.R., J. Koster, P. Mooyer, Gv. Gv. Noort , R. I. Kelley, W. R. Wilcox, R. J Wanders, R. C. Hennekam, J. C, Oosterwijk. 2003. Autosomal recessive HEM/Greenberg skeletal dysplasia is caused by 3 beta-hydroxysterol delta 14-reductase deficiency due to mutations in the lamin B receptor gene. Am J Hum Genet. 72(4):1013-7.
- Cohen, T.V., K. D Klarmann, K. Sakchaisri, J. P Cooper, D. Kuhns, M. Anver, P. F. Johnson, S. C. Williams, J. R. Keller, C. L. Stewart. 2008. The lamin B receptor under transcriptional control of C/EBPepsilon is required for morphological but not functional maturation of neutrophils. Hum Mol Genet. 17(19):2921-33.
If you publish research with this product, please let us know so we can cite your paper.

![Anti-Lamin B Receptor (LBR) [BBmLBR 12.F8] Antibody Anti-Lamin B Receptor (LBR) [BBmLBR 12.F8] Antibody](https://www.kerafast.com/MediaStorage/Product/Images/Medium/1759_2001202001270017580.jpg)
