Murine Osteocyte-like Cell Line MLO-Y4
MLO-Y4 cell line is useful to study effects of fluid flow and substrate stretching on activation of osteocyte-specific signaling pathways.
Highlights:
- Derived from a transgenic mouse in which the immortalizing T-antigen was expressed under control of the osteocalcin promoter
- High expression of osteocalcin
- High expression of connexin 43 and the antigen E11
- Retain a dendritic morphology, similar to that observed in primary osteocyte
Osteocytes are the most abundant bone cells in the body but also the most challenging to study because they are embedded in a mineralized matrix making them to difficult to isolate. This cell line MLO-Y4 makes it easier to study osteocyte function.
From the laboratory of Lynda Bonewald, PhD, University of Missouri - Kansas City.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
MLO-Y4 cell line is useful to study effects of fluid flow and substrate stretching on activation of osteocyte-specific signaling pathways.
Highlights:
- Derived from a transgenic mouse in which the immortalizing T-antigen was expressed under control of the osteocalcin promoter
- High expression of osteocalcin
- High expression of connexin 43 and the antigen E11
- Retain a dendritic morphology, similar to that observed in primary osteocyte
Osteocytes are the most abundant bone cells in the body but also the most challenging to study because they are embedded in a mineralized matrix making them to difficult to isolate. This cell line MLO-Y4 makes it easier to study osteocyte function.
From the laboratory of Lynda Bonewald, PhD, University of Missouri - Kansas City.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
| Product Type: | Cell Line |
| Name: | MLO-Y4 |
| Cell Type: | Osteocyte |
| Accession ID: | CVCL_M098 |
| Organism: | Mouse (female) |
| Morphology: | Dendritic morphology |
| Biosafety Level: | I |
| Subculturing: | Maintain stock cultures in complete culture medium at ~60-75% confluence on collagen coated plates at 37°C and at 5% CO 2. Passage at ~ 1:5 dilution (~1:7maximum) using 0.05% Trypsin/0.53 mM EDTA every 3-4 days (see Rosser J. et al. Methods Mol Biol. 2012;816:67-81 |
| Growth Conditions: | alpha-MEM, containing L-glutamine and deoxyribonucleosides, with penicillin/streptomycin at 100 U/ml and 2.5% FBS and 2.5% CS; both heat inactivated; Grown on dishes coated with [0.15 mg/ml] rat tail type 1 collagen (see Rosser J. et al. Methods Mol Biol. 2012;816:67-81). |
| Cryopreservation: | 60% alpha-MEM, 30% FBS, 10% DMSO, at 1 × 10 6cells/ml/cryovial (see Rosser J. et al. Methods Mol Biol. 2012;816:67-81) |
| Comments: | When first starting these cells, centrifuge at approx. 1,000 rpm, for 5–10 min. Aspirate media, gently resuspend and plate the cell pellet in media containing 5% FBS and 5% CS. This higher amount of serum, 10% total, is useful to give the cells an extra boost. The next day, check the viability of the cells. If there are a lot of fl oating “dead” cells, change the medium. You can revert back to medium with 2.5% FBS + 2.5% CS at this point. |
| Storage: | Liquid nitrogen |
| Shipped: | Dry ice |
MLO-Y4 Cells
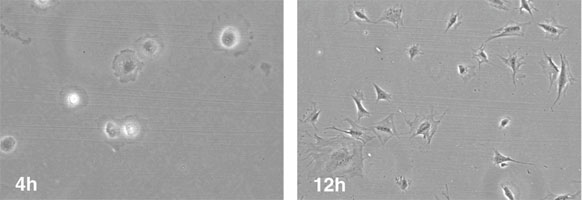
Appearance of the MLO-Y4 cells at 4 and 12 h after plating. The cells are attached by 4 h and already generate dendritic processes by 12 h.
Adapted from: Rosser J. et al. Methods Mol Biol. 2012;816:67-81.
- Kato Y, Windle J,Koop B, Qiao M, Bonewald L. Establishment of an osteocyte-like cell line, MLO-Y4 J Bone Min. Res. 12:2014-2023, 1997.
- Kato, Y, Boskey, A, Spevak, L, Dallas, M, Hori, M, Bonewald, LF, Establishment of an Osteoid Pre-Osteocyte like cell, MLO-A5, that spontaneously mineralizes in culture without the addition of beta-glycerol phosphate and ascorbic acid. J. Bone Min. Res. 16:1622-1633, 2001.
- Rosser J, Bonewald LF. Studying osteocyte function using the cell lines MLO-Y4 and MLO-A5. Methods Mol Biol. 2012;816:67-81.
- Sun Y, Kaur K, Kanayama K, Morinaga K, Park S, Hokugo A, Kozlowska A, McBride WH, Li J, Jewett A, Nishimura I. Plasticity of Myeloid Cells during Oral Barrier Wound Healing and the Development of Bisphosphonate-Related Osteonecrosis of the Jaw. J Biol Chem. 2016 Aug 11. pii: jbc.M116.735795. View Article
- Wang T, Han C, Tian P, Li PF, Ma XL. Role of Teriparatide in Glucocorticoid-induced Osteoporosis through Regulating Cellular Reactive Oxygen Species. Orthop Surg. 2018 May 10. View Article
- Kim HJ, Kim HJ, Choi Y, Bae MK, Hwang DS, Shin SH, Lee JY. Zoledronate Enhances Osteocyte-Mediated Osteoclast Differentiation by IL-6/RANKL Axis. Int J Mol Sci. 2019 Mar 22;20(6). pii: E1467. View Article
- Yamamoto H, Saito M, Goto T, Ueshima K, Ishida M, Hayashi S, Ikoma K, Mazda O, Kubo T. Heme oxygenase-1 prevents glucocorticoid and hypoxia-induced apoptosis and necrosis of osteocyte-like cells. Med Mol Morphol. 2019 Jan 31. View Article
- Parmentier L, Riffault M, Hoey DA. Utilizing Osteocyte Derived Factors to Enhance Cell Viability and Osteogenic Matrix Deposition within IPN Hydrogels. Materials (Basel). 2020;13(7):1690. Published 2020 Apr 4. View article
- Ueda S, Shimasaki M, Ichiseki T, Hirata H, Kawahara N, Ueda Y. Mitochondrial Transcription Factor A added to Osteocytes in a Stressed Environment has a Cytoprotective Effect. Int J Med Sci. 2020;17(9):1293-1299. Published 2020 May 23. View article
- Eichholz KF, Woods I, Riffault M, et al. Human bone marrow stem/stromal cell osteogenesis is regulated via mechanically activated osteocyte-derived extracellular vesicles [published online ahead of print, 2020 Jul 16]. Stem Cells Transl Med. 2020;10.1002/sctm.19-0405. View article
- Hirata H, Ueda S, Ichiseki T, Shimasaki M, Ueda Y, Kaneuji A, Kawahara N. Taurine Inhibits Glucocorticoid-Induced Bone Mitochondrial Injury, Preventing Osteonecrosis in Rabbits and Cultured Osteocytes. Int J Mol Sci. 2020 Sep 20;21(18):E6892. View article
- Wang Z, Weng Y, Ishihara Y, Odagaki N, Ei Hsu Hlaing E, Izawa T, Okamura H, Kamioka H. Loading history changes the morphology and compressive force-induced expression of receptor activator of nuclear factor kappa B ligand/osteoprotegerin in MLO-Y4 osteocytes. PeerJ. 2020 Nov 9;8:e10244. View article
If you publish research with this product, please let us know so we can cite your paper.