Human Deoxycytidine Kinase (dCK) WT
Recombinant full length (aa 1-260) wild-type Human Deoxycytidine kinase (dCK) purified by nickel-sepharose chromatography.
Highlights:
- Highly pure - >99% (SDS-PAGE)
- Phosphorylates physiological deoxyribonucleosides dC, dA, and dG
- Phosphorylates nucleoside analogs (e.g., AraC, gemcitabine, clofarabine, 3TC)
Deoxycytidine kinase (dCK, EC:2.7.1.74) is required for the phosphorylation of the deoxyribonucleosides deoxycytidine (dC), deoxyguanosine (dG), and deoxyadenosine (dA). dCK has a broad substrate specificity, and does not display selectivity based on the chirality of the substrate. It is also an essential enzyme for the phosphorylation of numerous nucleoside analogs widely employed as antiviral and chemotherapeutic agents.
Also available: Human Deoxycytidine Kinase (dCK) (R104M, D133A)
From the laboratory of Arnon Lavie, PhD, University of Illinois at Chicago.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
Recombinant full length (aa 1-260) wild-type Human Deoxycytidine kinase (dCK) purified by nickel-sepharose chromatography.
Highlights:
- Highly pure - >99% (SDS-PAGE)
- Phosphorylates physiological deoxyribonucleosides dC, dA, and dG
- Phosphorylates nucleoside analogs (e.g., AraC, gemcitabine, clofarabine, 3TC)
Deoxycytidine kinase (dCK, EC:2.7.1.74) is required for the phosphorylation of the deoxyribonucleosides deoxycytidine (dC), deoxyguanosine (dG), and deoxyadenosine (dA). dCK has a broad substrate specificity, and does not display selectivity based on the chirality of the substrate. It is also an essential enzyme for the phosphorylation of numerous nucleoside analogs widely employed as antiviral and chemotherapeutic agents.
Also available: Human Deoxycytidine Kinase (dCK) (R104M, D133A)
From the laboratory of Arnon Lavie, PhD, University of Illinois at Chicago.
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
| Catalog Number | Product | DataSheet | Size | AVAILABILITY | Price | Qty |
|---|
This product is for sale to Nonprofit customers only. For profit customers, please Contact Us for more information.
| Product Type: | Protein |
| Name: | WT human dCK |
| Accession ID: | P27707 |
| Source: | Recombinant expression in E. coli |
| Molecular Weight: | ~31 kDa |
| Amino Acid Sequence: | MATPPKRSCPSFSASSEGTRIKKISIEGNIAAGKSTFVNILKQLCEDWEVVPEPVARWCNVQSTQDEFEELTMSQKNGGNVLQMMYEKPERWSFTFQTYACLSRIRAQLASLNGKLKDAEKPVLFFERSVYSDRYIFASNLYESECMNETEWTIYQDWHDWMNNQFGQSLELDGIIYLQATPETCLHRIYLRGRNEEQGIPLEYLEKLHYKHESWLLHRTLKTNFDYLQEVPILTLDVNEDFKDKYESLVEKVKE |
| Fusion Tag(s): | N-terminal His tag |
| Purity: | >99% (SDS-PAGE) |
| Buffer: | 25 mM Hepes pH7.5, 200 mM NaCitrate, 10% glycerol, 5 mM DTT, 1 mM EDTA |
| Concentration: | 4.6mg/mL |
| Activity: | 6 IU/mg protein; kcat= 0.05/sec with dC as substrate; One unit of WT human dCK converts 1.0 µmole of dC and ATP to dCMP and ADP per minute at pH 7.5 at 37°C, as measured by a coupled enzyme system with 200 µM dC and 1 mM ATP. |
| Storage: | -80C |
| Shipped: | Dry ice |
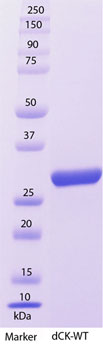
SDS-PAGE Analysis
- Sabini E, Ort S, Monnerjahn C, Konrad M, Lavie A. Structure of human dCK suggests strategies to improve anticancer and antiviral therapy. Nat Struct Biol. 2003 Jul;10(7):513-9.
- Sabini E, Hazra S, Konrad M, Lavie A. Nonenantioselectivity property of human deoxycytidine kinase explained by structures of the enzyme in complex with L- and D-nucleosides. J Med Chem. 2007 Jun 28;50(13):3004-14. Epub 2007 May 27.
If you publish research with this product, please let us know so we can cite your paper.


