Anti-Puromycin [3RH11] Antibody
This monoclonal antibody to puromycin provides a non-radioactive method to measure rates of global protein synthesis (mRNA translation) in cells or tissue slices incubated with puromycin, or animals treated with puromycin in vivo.
Highlights:
- Allows for the simple evaluation and quantification of translation directly using standard immunochemical methods
- Advantageous alternative to traditional pulse-chase methods, which rely on radioactive amino acid labeling
- Compatible with Western Blot and ELISA applications
Recombinant versions available from our sister company, Absolute Antibody:
- Manufactured using Absolute Antibody’s Recombinant Platform with variable regions (i.e., specificity) from the hybridoma 3RH11
Puromycin is an aminonucleoside antibiotic, derived from the Streptomyces alboniger bacterium, that causes premature chain termination during translation taking place in the ribosome. Part of the molecule resembles the 3' end of the aminoacylated tRNA, making it useful for protein translation analysis. Classical pulse-chase or flooding dose methods used to monitor protein synthesis rely on the measurement of radioactive methionine and cysteine labels. Analysis using puromycin immunodetection is an advantageous alternative to radioactive amino acid labeling, and allows for the evaluation/quantification of translation directly using standard immunochemical methods.
From the laboratory of Scot R. Kimball, PhD, Penn State College of Medicine.
Read related blog post, Puromycin Incorporation as a Measure of Global Protein Synthesis »
This monoclonal antibody to puromycin provides a non-radioactive method to measure rates of global protein synthesis (mRNA translation) in cells or tissue slices incubated with puromycin, or animals treated with puromycin in vivo.
Highlights:
- Allows for the simple evaluation and quantification of translation directly using standard immunochemical methods
- Advantageous alternative to traditional pulse-chase methods, which rely on radioactive amino acid labeling
- Compatible with Western Blot and ELISA applications
Recombinant versions available from our sister company, Absolute Antibody:
- Manufactured using Absolute Antibody’s Recombinant Platform with variable regions (i.e., specificity) from the hybridoma 3RH11
Puromycin is an aminonucleoside antibiotic, derived from the Streptomyces alboniger bacterium, that causes premature chain termination during translation taking place in the ribosome. Part of the molecule resembles the 3' end of the aminoacylated tRNA, making it useful for protein translation analysis. Classical pulse-chase or flooding dose methods used to monitor protein synthesis rely on the measurement of radioactive methionine and cysteine labels. Analysis using puromycin immunodetection is an advantageous alternative to radioactive amino acid labeling, and allows for the evaluation/quantification of translation directly using standard immunochemical methods.
From the laboratory of Scot R. Kimball, PhD, Penn State College of Medicine.
Read related blog post, Puromycin Incorporation as a Measure of Global Protein Synthesis »
| Catalog Number | Product | DataSheet | Size | AVAILABILITY | Price | Qty |
|---|
| Product Type: | Antibody |
| Name: | Anti-Puromycin (3RH11) |
| Host: | Mouse |
| Isotype: | see product name |
| Clonality: | Monoclonal |
| Clone Name: | 3RH11 |
| Specificity: | This antibody recognizes puromycin. |
| Immunogen: | puromycin hydrochloride |
| Format: | Liquid |
| Purification Method: | Protein G purified |
| Buffer: | PBS with 0.02% Proclin 300 |
| Tested Applications: | Western blotting (1:1,000), ELISA and Immunofluorescence microscopy. |
| Storage: | +4C (short-term), -20C (long-term); Avoid repeated freeze/thaw cycles. |
| Shipped: | Cold packs |
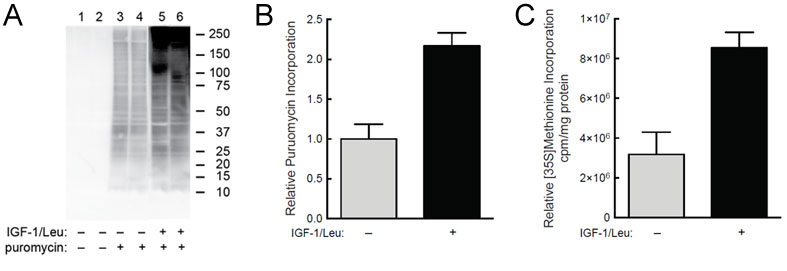
(A) C2C12 myoblasts were starved of serum and leucine for 2 hr and then IGF-1 and leucine were added to the medium of some of the cells for 45 min. Puromycin (1uM) was added to the medium of some of the cells (lanes 3-6) 30 min before harvest. (B) Quantification of western blot analysis from panel A. (C) In the same study, but in a separate set of culture dishes, cells were incubated with [35S]methionine instead of puromycin and incorporation was measured.
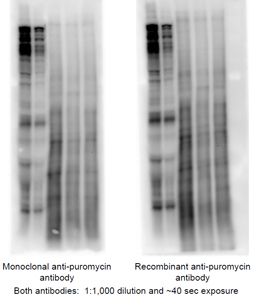
A Western blot was run using the same samples where one set was run on the left side of the gel and the other on the right. The left side was probed with our original monoclonal anti-puromycin antibody and the other side was probed with the recombinant anti-puromycin antibody, both at 1:1,000 dilution. The secondary antibody was used at the same dilution for both sides and they were both exposed for ~40 sec. The first two samples were from skeletal muscle of mice where the first lane is muscle from the control hindlimb and the other is from a hindlimb that had been immobilized with a cast for three days. The other 3 lanes are from HEK393T cells: the first lane is from cells incubated in complete medium, the middle lane is from cells incubated for 2 hr in medium lacking glucose and serum, and the last lane is cells incubated for 1.5 hr without glucose or serum and then glucose and serum were returned during the last 30 min. As expected, puromycin incorporation was lower in the immobilized hindlimb compared to the contralateral control hindlimb, and also in cells deprived of glucose and serum compared to cells in complete medium. Resupplementation partially restored incorporation.
Scot R. Kimball, PhD
Penn State College of Medicine
Puromycin inhibits protein synthesis, which some cells may be more or less sensitive to. It is therefore highly recommended that the concentration of puromycin for protein translation assays be optimized for cell type. The suggested concentration of puromycin is 1uM, but for cells that are more sensitive, lower concentrations of puromycin may give better results.
Because cell growth (and protein synthesis) slows dramatically as cell near confluence, it is recommended that experiments are performed during growth phase (40-50% confluence). Label with an optimized concentration of puromycin for at least 20-30 min.
- Templin AT, Maier B, Tersey SA, Hatanaka M, Mirmira RG. Maintenance of Pdx1 mRNA translation in islet β-cells during the unfolded protein response. Mol Endocrinol. 2014 Nov;28(11):1820-30. View Article
- Lee CS, Georgiou DK, Dagnino-Acosta A, Xu J, Ismailov II, Knoblauch M, Monroe TO, Ji R, Hanna AD, Joshi AD, Long C, Oakes J, Tran T, Corona BT, Lorca S, Ingalls CP, Narkar VA, Lanner JT, Bayle JH, Durham WJ, Hamilton SL. Ligands for FKBP12 increase Ca2+ influx and protein synthesis to improve skeletal muscle function. J Biol Chem. 2014 Sep 12;289(37):25556-70. View Article
- Steiner JL, Crowell KT, Kimball SR, Lang CH. Disruption of REDD1 gene ameliorates sepsis-induced decrease in mTORC1 signaling but has divergent effects on proteolytic signaling in skeletal muscle. Am J Physiol Endocrinol Metab. 2015 Dec 15;309(12):E981-94. View Article
- Lee KH, Zhang P, Kim HJ, Mitrea DM, Sarkar M, Freibaum BD, Cika J, Coughlin M, Messing J, Molliex A, Maxwell BA, Kim NC, Temirov J, Moore J, Kolaitis RM, Shaw TI, Bai B, Peng J, Kriwacki RW, Taylor JP. C9orf72 Dipeptide Repeats Impair the Assembly, Dynamics, and Function of Membrane-Less Organelles. Cell. 2016 Oct 20;167(3):774-788.e17. View Article
- Mirzoev T, Tyganov S, Vilchinskaya N, Lomonosova Y, Shenkman B. Key Markers ofmTORC1-Dependent and mTORC1-Independent Signaling Pathways Regulating ProteinSynthesis in Rat Soleus Muscle During Early Stages of Hindlimb Unloading. CellPhysiol Biochem. 2016 Aug 19;39(3):1011-1020. View Article
- Wang Y, Krais JJ, Bernhardy AJ, Nicolas E, Cai KQ, Harrell MI, Kim HH, George E, Swisher EM, Simpkins F, Johnson N. RING domain-deficient BRCA1 promotes PARPinhibitor and platinum resistance. J Clin Invest. 2016 Aug 1;126(8):3145-57. View Article
- Suzuki Y, Chin WX, Han Q, Ichiyama K, Lee CH, Eyo ZW, Ebina H, Takahashi H,Takahashi C, Tan BH, Hishiki T, Ohba K, Matsuyama T, Koyanagi Y, Tan YJ, SawasakiT, Chu JJ, Vasudevan SG, Sano K, Yamamoto N. Characterization of RyDEN (C19orf66)as an Interferon-Stimulated Cellular Inhibitor against Dengue Virus Replication. PLoS Pathog. 2016 Jan 6;12(1):e1005357. View Article
- Wang XJ, Yang X, Wang RX, Jiao HC, Zhao JP, Song ZG, Lin H. Leucine alleviates dexamethasone-induced suppression of muscle protein synthesis via synergy involvement of mTOR and AMPK pathways. Biosci Rep. 2016 Jun 17;36(3). pii:e00346. View Article
- Wang R, Jiao H, Zhao J, Wang X, Lin H. Glucocorticoids Enhance MuscleProteolysis through a Myostatin-Dependent Pathway at the Early Stage. PLoS One.2016 May 26;11(5):e0156225. View Article
- Kainulainen M, Lau S, Samuel CE, Hornung V, Weber F. NSs virulence factor ofRift Valley fever virus engages the F-box proteins FBXW11 and ?-TRCP1 to degrade the antiviral protein kinase PKR. J Virol. 2016 Apr 27. pii: JVI.00016-16. View Article
- Vu?i?evi? D, Gehre M, Dhamija S, Friis-Hansen L, Meierhofer D, Sauer S, Ørom UA. The long non-coding RNA PARROT is an upstream regulator of c-Myc and affects proliferation and translation. Oncotarget. 2016 Apr 5. View Article
- Gordon BS, Liu C, Steiner JL, Nader GA, Jefferson LS, Kimball SR. Loss of REDD1 augments the rate of the overload-induced increase in muscle mass. Am J Physiol Regul Integr Comp Physiol. 2016 Sep 1;311(3):R545-57. View Article
- Ueki N, Wang W, Swenson C, McNaughton C, Sampson NS, Hayman MJ. Synthesis and Preclinical Evaluation of a Highly Improved Anticancer Prodrug Activated by Histone Deacetylases and Cathepsin L. Theranostics 2016; 6(6):808-816. doi:10.7150/thno.13826. View Article
- J.J. David Ho, Miling Wang, Timothy E. Audas, Deukwoo Kwon, Steven K. Carlsson, Sara Timpano, Sonia L. Evagelou, Shaun Brothers, Mark L. Gonzalgo, Jonathan R. Krieger, Steven Chen, James Uniacke, Stephen Lee. Systemic Reprogramming of Translation Efficiencies on Oxygen Stimulus. Cell Reports. DOI: http://dx.doi.org/10.1016/j.celrep.2016.01.036. View Article
- Khoutorsky A, Bonin RP, Sorge RE, Gkogkas CG, Pawlowski SA, Jafarnejad SM, Pitcher MH, Alain T, Perez-Sanchez J, Salter EW, Martin L, Ribeiro-da-Silva A, De Koninck Y, Cervero F, Mogil JS, Sonenberg N. Translational control of nociception via 4E-binding protein 1. Elife. 2015 Dec 18;4. View Article
- Di Salvio M, Piccinni V, Gerbino V, Mantoni F, Camerini S, Lenzi J, Rosa A, Chellini L, Loreni F, Carrì MT, Bozzoni I, Cozzolino M, Cestra G. Pur-alpha functionally interacts with FUS carrying ALS-associated mutations. Cell Death Dis. 2015 Oct 22;6:e1943. View Article
- Steiner JL, Gordon BS, Lang CH. Moderate alcohol consumption does not impair overload-induced muscle hypertrophy and protein synthesis. Physiol Rep. 2015 Mar;3(3). pii: e12333. doi: 10.14814/phy2.12333. View Article
- Zhang Q, Joshi SK, Lovett DH, Zhang B, Bodine S, Kim HT, Liu X. Matrix metalloproteinase-2 plays a critical role in overload induced skeletal muscle hypertrophy. Muscles Ligaments Tendons J. 2015 Feb 5;4(4):446-54. View Article
- Hayasaka M, Tsunekawa H, Yoshinaga M, Murakami T. Endurance exercise induces REDD1 expression and transiently decreases mTORC1 signaling in rat skeletal muscle. Physiol Rep. 2014 Dec 24;2(12). pii: e12254. doi: 10.14814/phy2.12254. View Article
- Kelleher AR, Kimball, SR, Dennis MD, Schilder RJ and Jefferson LS. The mTORC1 signaling repressors REDD1/2 are rapidly induced and activation of p70S6K1 by leucine is defective in skeletal muscle of an immobilized rat hindlimb. Am J Physiol Endocrinol Metab. 304(2):E229-236. 2013.
- Yasuda K, Zhang H, Loiselle D, Haystead T, Macara IG, Mili S. The RNA-binding protein Fus directs translation of localized mRNAs in APC-RNP granules. J Cell Biol. 2013 Dec 9;203(5):737-46.
- Dai N, Christiansen J, Nielsen FC, Avruch J. mTOR complex 2 phosphorylates IMP1 cotranslationally to promote IGF2 production and the proliferation of mouse embryonic fibroblasts. Genes Dev. 2013 Feb 1;27(3):301-12.
- Belozerov VE, Ratkovic S, McNeill H, Hilliker AJ, McDermott JC. In vivo interaction proteomics reveal a novel p38MAPK/Rack1 pathway regulating proteostasis in Drosophila muscle. Mol Cell Biol. 2013 Nov 25.
- Ueki N, Lee S, Sampson NS, Hayman MJ. Selective cancer targeting with prodrugs activated by histone deacetylases and a tumour-associated protease. Nat Commun. 2013;4:2735.
- Ha SW, Ju D, Xie Y. Nuclear import factor Srp1 and its associated protein Sts1 couple ribosome-bound nascent polypeptides to proteasomes for cotranslational degradation. J Biol Chem. 2013 Dec 12.
- Naydenov NG, Baranwal S, Khan S, Feygin A, Gupta P, Ivanov AI. Novel mechanism of cytokine-induced disruption of epithelial barriers Janus kinase and protein kinase D-dependent downregulation of junction protein expression. Tissue Barriers 1:4, e25231.
- Park YS, Liu Z, Vasamsetti BM, Cho NJ. The ERK1/2 and mTORC1 Signaling Pathways Are Involved in the Muscarinic Acetylcholine Receptor-Mediated Proliferation of SNU-407 Colon Cancer Cells. J Cell Biochem. 2016 Dec;117(12):2854-2863. doi: 10.1002/jcb.25597. PubMed PMID: 27167250. View Article
- Crowell KT, Steiner JL, Coleman CS, Lang CH. Decreased Whole-Body Fat Mass Produced by Chronic Alcohol Consumption is Associated with Activation of S6K1-Mediated Protein Synthesis and Increased Autophagy in Epididymal White Adipose Tissue. Alcohol Clin Exp Res. 2016 Sep;40(9):1832-45. doi: 10.1111/acer.13159. PubMed PMID: 27464336; PubMed Central PMCID: PMC5009010. View Article
- Spangler B, Morgan CW, Fontaine SD, Vander Wal MN, Chang CJ, Wells JA, Renslo AR. A reactivity-based probe of the intracellular labile ferrous iron pool. Nat Chem Biol. 2016 Sep;12(9):680-5. doi: 10.1038/nchembio.2116. PubMed PMID: 27376690; PubMed Central PMCID: PMC4990480. View Article
- Steiner JL, Fukuda DH, Rossetti ML, Hoffman JR, Gordon BS. Castration Alters Protein Balance Following High Frequency Muscle Contraction. J Appl Physiol (1985). 2016 Dec 1:jap.00740.2016. doi: 10.1152/japplphysiol.00740.2016. [Epub ahead of print] PubMed PMID: 27909227. View Article
- Reid DW, Tay AS, Sundaram JR, Lee IC, Chen Q, George SE, Nicchitta CV, Shenolikar S. Complementary Roles of GADD34- and CReP-Containing Eukaryotic Initiation Factor 2α Phosphatases during the Unfolded Protein Response. Mol Cell Biol. 2016 Jun 15;36(13):1868-80. doi: 10.1128/MCB.00190-16. PubMed PMID: 27161320; PubMed Central PMCID: PMC4911741. View Article
- Stretton C, Hoffmann TM, Munson MJ, Prescott A, Taylor PM, Ganley IG, Hundal HS. GSK3-mediated raptor phosphorylation supports amino-acid-dependent mTORC1-directed signalling. Biochem J. 2015 Sep 1;470(2):207-21. doi: 10.1042/BJ20150404. PubMed PMID: 26348909; PubMed Central PMCID: PMC4652938. View Article
- Fay A, Glickman MS. An essential nonredundant role for mycobacterial DnaK in native protein folding. PLoS Genet. 2014 Jul 24;10(7):e1004516. doi: 10.1371/journal.pgen.1004516. PubMed PMID: 25058675; PubMed Central PMCID: PMC4109909. View Article
- Cattin ME, Wang J, Weldrick JJ, Roeske CL, Mak E, Thorn SL, DaSilva JN, Wang Y, Lusis AJ, Burgon PG. Deletion of MLIP (muscle-enriched A-type lamin-interacting protein) leads to cardiac hyperactivation of Akt/mammalian target of rapamycin (mTOR) and impaired cardiac adaptation. J Biol Chem. 2015 Oct 30;290(44):26699-714. doi: 10.1074/jbc.M115.678433. PubMed PMID: 26359501; PubMed Central PMCID: PMC4646324. (supplemental info) View Article
- Pirinen E, Cantó C, Jo YS, Morato L, Zhang H, Menzies KJ, Williams EG, Mouchiroud L, Moullan N, Hagberg C, Li W, Timmers S, Imhof R, Verbeek J, Pujol A, van Loon B, Viscomi C, Zeviani M, Schrauwen P, Sauve AA, Schoonjans K, Auwerx J. Pharmacological Inhibition of poly(ADP-ribose) polymerases improves fitness and mitochondrial function in skeletal muscle. Cell Metab. 2014 Jun 3;19(6):1034-41. doi: 10.1016/j.cmet.2014.04.002. PubMed PMID: 24814482; PubMed Central PMCID: PMC4047186. View Article
- Lu Y, Liang FX, Wang X. A synthetic biology approach identifies the mammalian UPR RNA ligase RtcB. Mol Cell. 2014 Sep 4;55(5):758-70. doi: 10.1016/j.molcel.2014.06.032. PubMed PMID: 25087875; PubMed Central PMCID: PMC4156904. View Article
- Lin F, Marcelo KL, Rajapakshe K, Coarfa C, Dean A, Wilganowski N, Robinson H, Sevick E, Bissig KD, Goldie LC, Means AR, York B. The camKK2/camKIV relay is an essential regulator of hepatic cancer. Hepatology. 2015 Aug;62(2):505-20. doi: 10.1002/hep.27832. PubMed PMID: 25847065; PubMed Central PMCID: PMC4515151. View Article
- Watson A, Lipina C, McArdle HJ, Taylor PM, Hundal HS. Iron depletion suppresses mTORC1-directed signalling in intestinal Caco-2 cells via induction of REDD1. Cell Signal. 2016 May;28(5):412-24. doi: 10.1016/j.cellsig.2016.01.014. PubMed PMID: 26827808; PubMed Central PMCID: PMC4804389. View Article
- Shiina N, Nakayama K. RNA granule assembly and disassembly modulated by nuclear factor associated with double-stranded RNA 2 and nuclear factor 45. J Biol Chem. 2014 Jul 25;289(30):21163-80. PubMed PMID: 24920670; PubMed Central PMCID: PMC4110319. View Article
- Ogasawara R, Sato K, Matsutani K, Nakazato K, Fujita S. The order of concurrent endurance and resistance exercise modifies mTOR signaling and protein synthesis in rat skeletal muscle. Am J Physiol Endocrinol Metab. 2014 May 15;306(10):E1155-62. doi: 10.1152/ajpendo.00647.2013. PubMed PMID: 24691029. View Article
- Sethna F, Feng W, Ding Q, Robison AJ, Feng Y, Wang H. Enhanced expression of ADCY1 underlies aberrant neuronal signalling and behaviour in a syndromic autism model. Nat Commun. 2017 Feb 20;8:14359. doi: 10.1038/ncomms14359. PubMed PMID: 28218269; PubMed Central PMCID: PMC5321753. View Article
- Dwyer JM, Maldonado-Avilés JG, Lepack AE, DiLeone RJ, Duman RS. Ribosomal protein S6 kinase 1 signaling in prefrontal cortex controls depressive behavior. Proc Natl Acad Sci U S A. 2015 May 12;112(19):6188-93. doi: 10.1073/pnas.1505289112. Epub 2015 Apr 27. PubMed PMID: 25918363; PubMed Central PMCID: PMC4434715. View Article
- Lipton JO, Yuan ED, Boyle LM, Ebrahimi-Fakhari D, Kwiatkowski E, Nathan A, Güttler T, Davis F, Asara JM, Sahin M. The Circadian Protein BMAL1 Regulates Translation in Response to S6K1-Mediated Phosphorylation. Cell. 2015 May 21;161(5):1138-51. doi: 10.1016/j.cell.2015.04.002. Epub 2015 May 14. PubMed PMID: 25981667; PubMed Central PMCID: PMC4447213 View Article
- Nie D, Chen Z, Ebrahimi-Fakhari D, Di Nardo A, Julich K, Robson VK, Cheng YC, Woolf CJ, Heiman M, Sahin M. The Stress-Induced Atf3-Gelsolin Cascade Underlies Dendritic Spine Deficits in Neuronal Models of Tuberous Sclerosis Complex. J Neurosci. 2015 Jul 29;35(30):10762-72. View Article
- Potts MB, McMillan EA, Rosales TI, Kim HS, Ou YH, Toombs JE, Brekken RA, Minden MD, MacMillan JB, White MA. Mode of action and pharmacogenomic biomarkers for exceptional responders to didemnin B. Nat Chem Biol. 2015 Jun;11(6):401-8. View Article
- Tom Dieck S, Kochen L, Hanus C, Heumüller M, Bartnik I, Nassim-Assir B, Merk K, Mosler T, Garg S, Bunse S, Tirrell DA, Schuman EM. Direct visualization of newly synthesized target proteins in situ. Nat Methods. 2015 May;12(5):411-4. View Article
- Miyake M, Nomura A, Ogura A, Takehana K, Kitahara Y, Takahara K, Tsugawa K, Miyamoto C, Miura N, Sato R, Kurahashi K, Harding HP, Oyadomari M, Ron D, Oyadomari S. Skeletal muscle-specific eukaryotic translation initiation factor 2α phosphorylation controls amino acid metabolism and fibroblast growth factor 21-mediated non-cell-autonomous energy metabolism. FASEB J. 2016 Feb;30(2):798-812. View Article
- Su KH, Cao J, Tang Z, Dai S, He Y, Sampson SB, Benjamin IJ, Dai C. HSF1 critically attunes proteotoxic stress sensing by mTORC1 to combat stress and promote growth. Nat Cell Biol. 2016 May;18(5):527-39. View Article
- Lee CS, Hanna AD, Wang H, Dagnino-Acosta A, Joshi AD, Knoblauch M, Xia Y, Georgiou DK, Xu J, Long C, Amano H, Reynolds C, Dong K, Martin JC, Lagor WR, Rodney GG, Sahin E, Sewry C, Hamilton SL. A chemical chaperone improves muscle function in mice with a RyR1 mutation. Nat Commun. 2017 Mar 24;8:14659. View Article
- Crowell KT, Soybel DI, Lang CH. Restorative Mechanisms Regulating Protein Balance in Skeletal Muscle During Recovery From Sepsis. Shock. 2017 Apr;47(4):463-473. View Article
- McLean KJ, Jacobs-Lorena M. Plasmodium falciparum Maf1 Confers Survival upon Amino Acid Starvation. MBio. 2017 Mar 28;8(2). pii: e02317-16. View Article
- Irena Vlatkovic, Sivakumar Sambandan, Georgi Tushev, Mantian Wang, Irina Epstein, Caspar Glock, Nicole Fuerst, Ivan Cajigas, Erin Schuman. Poly(A) Binding Protein Nuclear 1 regulates the polyadenylation of key synaptic plasticity genes and plays a role in homeostatic plasticity. bioRxiv. doi: https://doi.org/10.1101/121194. View Article
- Liu TY, Huang HH, Wheeler D, Xu Y, Wells JA, Song YS, Wiita AP. Time-Resolved Proteomics Extends Ribosome Profiling-Based Measurements of Protein Synthesis Dynamics. Cell Syst. 2017 Jun 28;4(6):636-644.e9. View Article
- Mirzoev T, Tyganov S, Petrova I, Gnyubkin V, Laroche N, Vico L, Shenkman B. Divergent Anabolic Signalling responses of Murine Soleus and Tibialis Anterior Muscles to Chronic 2G Hypergravity. Sci Rep. 2017 Jun 14;7(1):3514. View Article
- Ogami K, Richard P, Chen Y, Hoque M, Li W, Moresco JJ, Yates JR 3rd, Tian B, Manley JL. An Mtr4/ZFC3H1 complex facilitates turnover of unstable nuclear RNAs to prevent their cytoplasmic transport and global translational repression. Genes Dev. 2017 Jun 15;31(12):1257-1271. View Article
- Hatanaka M, Anderson-Baucum E, Lakhter A, Kono T, Maier B, Tersey SA, Tanizawa Y, Evans-Molina C, Mirmira RG, Sims EK. Chronic high fat feeding restricts islet mRNA translation initiation independently of ER stress via DNA damage and p53 activation. Sci Rep. 2017 Jun 19;7(1):3758. View Article
- Crowell KT, Soybel DI, Lang CH. Inability to replete white adipose tissue during recovery phase of sepsis is associated with increased autophagy, apoptosis, and proteasome activity. Am J Physiol Regul Integr Comp Physiol. 2017 Mar 1;312(3):R388-R399. View Article
- Pal R, Bondar VV, Adamski CJ, Rodney GG, Sardiello M. Inhibition of ERK1/2 Restores GSK3β Activity and Protein Synthesis Levels in a Model of Tuberous Sclerosis. Sci Rep. 2017 Jun 23;7(1):4174. View Article
- Lipton JO, Boyle LM, Yuan ED, Hochstrasser KJ, Chifamba FF, Nathan A, Tsai PT, Davis F, Sahin M. Aberrant Proteostasis of BMAL1 Underlies Circadian Abnormalities in a Paradigmatic mTOR-opathy. Cell Rep. 2017 Jul 25;20(4):868-880. View Article
- Krause AR, Speacht TL, Zhang Y, Lang CH, Donahue HJ. Simulated space radiation sensitizes bone but not muscle to the catabolic effects of mechanical unloading. PLoS One. 2017 Aug 2;12(8):e0182403. View Article
- Deliu LP, Ghosh A, Grewal SS. Investigation of protein synthesis in Drosophila larvae using puromycin labelling. Biol Open. 2017 Aug 15;6(8):1229-1234. View Article
- Zappulo A, van den Bruck D, Ciolli Mattioli C, Franke V, Imami K, McShane E, Moreno-Estelles M, Calviello L, Filipchyk A, Peguero-Sanchez E, Müller T, Woehler A, Birchmeier C, Merino E, Rajewsky N, Ohler U, Mazzoni EO, Selbach M, Akalin A, Chekulaeva M. RNA localization is a key determinant of neurite-enriched proteome. Nat Commun. 2017 Sep 19;8(1):583. View Article
- Kelly E, Schaeffer SM, Dhamne SC, Lipton JO, Lindemann L, Honer M, Jaeschke G, Super CE, Lammers SH, Modi ME, Silverman JL, Dreier JR, Kwiatkowski DJ, Rotenberg A, Sahin M. mGluR5 Modulation of Behavioral and Epileptic Phenotypes in a Mouse Model of Tuberous Sclerosis Complex. Neuropsychopharmacology. 2017 Dec 5. View Article
- Camarena V, Sant DW, Huff TC, Mustafi S, Muir RK, Aron AT, Chang CJ, Renslo AR, Monje PV, Wang G. cAMP signaling regulates DNA hydroxymethylation by augmenting the intracellular labile ferrous iron pool. Elife. 2017 Dec 14;6. pii: e29750. View Article
- Sriskanthadevan-Pirahas S, Deshpande R, Lee B, Grewal SS. Ras/ERK-signalling promotes tRNA synthesis and growth via the RNA polymerase III repressor Maf1 in Drosophila. PLoS Genet. 2018 Feb 5;14(2):e1007202. View Article
- Farley-Barnes KI, McCann KL, Ogawa LM, Merkel J, Surovtseva YV, Baserga SJ. Diverse Regulators of Human Ribosome Biogenesis Discovered by Changes in Nucleolar Number. Cell Rep. 2018 Feb 13;22(7):1923-1934. View Article
- DeBoer ML, Martinson KM, Pampusch MS, Hansen AM, Wells SM, Ward C, Hathaway M. Cultured equine satellite cells as a model system to assess leucine stimulated protein synthesis in horse muscle. J Anim Sci. 2018 Feb 15;96(1):143-153. View Article
- Kapadia B, Nanaji NM, Bhalla K, Bhandary B, Lapidus R, Beheshti A, Evens AM, Gartenhaus RB. Fatty Acid Synthase induced S6Kinase facilitates USP11-eIF4B complex formation for sustained oncogenic translation in DLBCL. Nat Commun. 2018 Feb 26;9(1):829. View Article
- Zamurrad S, Hatch HAM, Drelon C, Belalcazar HM, Secombe J. A Drosophila Model of Intellectual Disability Caused by Mutations in the Histone Demethylase KDM5. Cell Rep. 2018 Feb 27;22(9):2359-2369. View Article
- Maity S, Rah S, Sonenberg N, Gkogkas CG, Nguyen PV. Norepinephrine triggers metaplasticity of LTP by increasing translation of specific mRNAs. Learn Mem. 2015 Sep 15;22(10):499-508. View Article
- Gantois I, Khoutorsky A, Popic J, Aguilar-Valles A, Freemantle E, Cao R, Sharma V, Pooters T, Nagpal A, Skalecka A, Truong VT, Wiebe S, Groves IA, Jafarnejad SM, Chapat C, McCullagh EA, Gamache K, Nader K, Lacaille JC, Gkogkas CG, Sonenberg N. Metformin ameliorates core deficits in a mouse model of fragile X syndrome. Nat Med. 2017 Jun;23(6):674-677.View Article
- Miki H, Nakamura S, Oda A, Tenshin H, Teramachi J, Hiasa M, Bat-Erdene A, Maeda Y, Oura M, Takahashi M, Iwasa M, Harada T, Fujii S, Kurahashi K, Yoshida S, Kagawa K, Endo I, Aihara K, Ikuo M, Itoh K, Hayashi K, Nakamura M, Abe M. Effective impairment of myeloma cells and their progenitors by hyperthermia. Oncotarget. 2017 Dec 7;9(12):10307-10316. View Article
- Fujii S, Nakamura S, Oda A, Miki H, Tenshin H, Teramachi J, Hiasa M, Bat-Erdene A, Maeda Y, Oura M, Takahashi M, Iwasa M, Endo I, Yoshida S, Aihara KI, Kurahashi K, Harada T, Kagawa K, Nakao M, Sano S, Abe M. Unique anti-myeloma activity by thiazolidine-2,4-dione compounds with Pim inhibiting activity. Br J Haematol. 2018 Jan;180(2):246-258. View Article
- Kaur H, He B, Zhang C, Rodriguez E, Hage DS, Moreau R. Piperine potentiates curcumin-mediated repression of mTORC1 signaling in human intestinal epithelial cells: implications for the inhibition of protein synthesis and TNFα signaling. J Nutr Biochem. 2018 Apr 25;57:276-286. View Article
- Hoshi O, Sugizaki A, Cho Y, Takei N. BDNF Reduces eEF2 Phosphorylation and Enhances Novel Protein Synthesis in the Growth Cones of Dorsal Root Ganglia Neurons. Neurochem Res. 2018 May 7. View Article
- Wang R, Jiao H, Zhao J, Wang X, Lin H. L-Arginine Enhances Protein Synthesis by Phosphorylating mTOR (Thr 2446) in a Nitric Oxide-Dependent Manner in C2C12 Cells. Oxid Med Cell Longev. 2018 Apr 26;2018:7569127. View Article
- Drelon C, Belalcazar HM, Secombe J. The Histone Demethylase KDM5 Is Essential for Larval Growth in Drosophila. Genetics. 2018 Jul;209(3):773-787. View Article
- Singh RK, Kolonin AM, Fiorotto ML, Cooper TA. Rbfox-Splicing Factors Maintain Skeletal Muscle Mass by Regulating Calpain3 and Proteostasis. Cell Rep. 2018 Jul 3;24(1):197-208. View Article
- Kawata K, Hatano A, Yugi K, Kubota H, Sano T, Fujii M, Tomizawa Y, Kokaji T, Tanaka KY, Uda S, Suzuki Y, Matsumoto M, Nakayama KI, Saitoh K, Kato K, Ueno A, Ohishi M, Hirayama A, Soga T, Kuroda S. Trans-omic Analysis Reveals Selective Responses to Induced and Basal Insulin across Signaling, Transcriptional, and Metabolic Networks. iScience. 2018 Sep 28;7:212-229. View Article
- Shimkus KL, Jefferson LS, Gordon BS, Kimball SR. Repressors of mTORC1 act to blunt the anabolic response to feeding in the soleus muscle of a cast-immobilized mouse hindlimb. Physiol Rep. 2018 Oct;6(20):e13891. View Article
- Lang CH. Lack of sexual dimorphism on the inhibitory effect of alcohol on muscle protein synthesis in rats under basal conditions and after anabolic stimulation. Physiol Rep. 2018 Dec;6(23):e13929. View Article
- Steiner JL, Lang CH. Ethanol acutely antagonizes the refeeding-induced increase in mTOR-dependent protein synthesis and decrease in autophagy in skeletal muscle. Mol Cell Biochem. 2018 Dec 6. View Article
- Ciolli Mattioli C, Rom A, Franke V, Imami K, Arrey G, Terne M, Woehler A, Akalin A, Ulitsky I, Chekulaeva M. Alternative 3' UTRs direct localization of functionally diverse protein isoforms in neuronal compartments. Nucleic Acids Res. 2018 Dec 22. View Article
- Zadra G, Ribeiro CF, Chetta P, Ho Y, Cacciatore S, Gao X, Syamala S, Bango C, Photopoulos C, Huang Y, Tyekucheva S, Bastos DC, Tchaicha J, Lawney B, Uo T, D'Anello L, Csibi A, Kalekar R, Larimer B, Ellis L, Butler LM, Morrissey C, McGovern K, Palombella VJ, Kutok JL, Mahmood U, Bosari S, Adams J, Peluso S, Dehm SM, Plymate SR, Loda M. Inhibition of de novo lipogenesis targets androgen receptor signaling in castration-resistant prostate cancer. Proc Natl Acad Sci U S A. 2019 Jan 8;116(2):631-640. View Article
- Shiina N. Liquid- and solid-like RNA granules form through specific scaffold proteins and combine into biphasic granules. J Biol Chem. 2019 Jan 3. View Article
- Lee JS, Lee H, Lee S, Kang JH, Lee SH, Kim SG, Cho ES, Kim NH, Yook JI, Kim SY. Loss of SLC25A11 causes suppression of NSCLC and melanoma tumor formation. EBioMedicine. 2019 Feb;40:184-197. View Article
- Tyganov S, Mirzoev T, Shenkman B. An Anabolic Signaling Response of Rat Soleus Muscle to Eccentric Contractions Following Hindlimb Unloading: A Potential Role of Stretch-Activated Ion Channels. Int J Mol Sci. 2019 Mar 7;20(5). pii: E1165. View Article
- Wang R, Li K, Wang H, Jiao H, Wang X, Zhao J, Lin H. Endogenous CSE/Hydrogen Sulfide System Regulates the Effects of Glucocorticoids and Insulin on Muscle Protein Synthesis. Oxid Med Cell Longev. 2019 Apr 7;2019:9752698. View Article
- O'Reilly C, Cho JH, Qi Q, Peters JL, Fukuda Y, Frase S, Peng J, Schuetz JD, Cheng Y, Yoon SO, Han MJ. Metabolic switching in pluripotent stem cells reorganizes energy metabolism and subcellular organelles. Exp Cell Res. 2019 Jun 1;379(1):55-64. View Article
- Mekheal M, Steiner JL, Lang CH. Acute alcohol prevents the refeeding-induced decrease in autophagy but does not alter the increased protein synthetic response in heart. Alcohol. 2018 Dec;73:79-88. View Article
- Zhang M, Wu Y, Wang M, Wang Y, Tausif R, Yang Y. Genistein rescues hypoxia-induced pulmonary arterial hypertension through estrogen receptor and β-adrenoceptor signaling. J Nutr Biochem. 2018 Aug;58:110-118. View Article
- Umegaki Y, Brotons AM, Nakanishi Y, Luo Z, Zhang H, Bonni A, Ikeuchi Y. Palladin Is a Neuron-Specific Translational Target of mTOR Signaling That Regulates Axon Morphogenesis. J Neurosci. 2018 May 23;38(21):4985-4995. View Article
- Reineke LC, Cheema SA, Dubrulle J, Neilson JR. Chronic starvation induces noncanonical pro-death stress granules. J Cell Sci. 2018 Oct 5;131(19). pii: jcs220244. View Article
- DeBoer ML, Martinson KL, Kuhle KJ, Sheaffer CC, Hathaway MR. Plasma Amino Acid Concentrations of Horses Grazing Alfalfa, Cool-Season Perennial Grasses, and Teff. J Equine Vet Sci. 2019 Jan;72:72-78. View Article
- Rangaraju V, Lauterbach M, Schuman EM. Spatially Stable Mitochondrial Compartments Fuel Local Translation during Plasticity. Cell. 2019 Jan 10;176(1-2):73-84.e15.View Article
- Wu R, Dang F, Li P, Wang P, Xu Q, Liu Z, Li Y, Wu Y, Chen Y, Liu Y. The Circadian Protein Period2 Suppresses mTORC1 Activity via Recruiting Tsc1 to mTORC1 Complex. Cell Metab. 2019 Mar 5;29(3):653-667.e6. View Article
- Yik-Sham Chung C, Timblin GA, Saijo K, Chang CJ. Versatile Histochemical Approach to Detection of Hydrogen Peroxide in Cells and Tissues Based on Puromycin Staining. J Am Chem Soc. 2018 May 16;140(19):6109-6121. View Article
- Chen R, Park HA, Mnatsakanyan N, Niu Y, Licznerski P, Wu J, Miranda P, Graham M, Tang J, Boon AJW, Cossu G, Mandemakers W, Bonifati V, Smith PJS, Alavian KN, Jonas EA. Parkinson's disease protein DJ-1 regulates ATP synthase protein components to increase neuronal process outgrowth. Cell Death Dis. 2019 Jun 13;10(6):469. View Article
- Fritzlar S, Aktepe TE, Chao YW, Kenney ND, McAllaster MR, Wilen CB, White PA, Mackenzie JM. Mouse Norovirus Infection Arrests Host Cell Translation Uncoupled from the Stress Granule-PKR-eIF2α Axis. MBio. 2019 Jun 18;10(3). pii: e00960-19. View Article
- Morrissey JA, Mockett BG, Singh A, Kweon D, Ohline SM, Tate WP, Hughes SM, Abraham WC. A C-terminal peptide from secreted amyloid precursor protein-α enhances long-term potentiation in rats and a transgenic mouse model of Alzheimer's disease. Neuropharmacology. 2019 Jun 13;157:107670. View Article
- Morriss GR, Rajapakshe K, Huang S, Coarfa C, Cooper TA. Mechanisms of skeletal muscle wasting in a mouse model for myotonic dystrophy type 1. Hum Mol Genet. 2018 Aug 15;27(16):2789-2804. View Article
- Elamri I, Heumüller M, Herzig LM, Stirnal E, Wachtveitl J, Schuman EM, Schwalbe H. A New Photocaged Puromycin for an Efficient Labeling of Newly Translated Proteins in Living Neurons. Chembiochem. 2018 Dec 4;19(23):2458-2464. View Article
- Megat S, Ray PR, Moy JK, Lou TF, Barragán-Iglesias P, Li Y, Pradhan G, Wanghzou A, Ahmad A, Burton MD, North RY, Dougherty PM, Khoutorsky A, Sonenberg N, Webster KR, Dussor G, Campbell ZT, Price TJ. Nociceptor Translational Profiling Reveals the Ragulator-Rag GTPase Complex as a Critical Generator of Neuropathic Pain. J Neurosci. 2019 Jan 16;39(3):393-411. View Article
- Mirzoev TM, Tyganov SA, Petrova IO, Shenkman BS. Acute recovery from disuse atrophy: the role of stretch-activated ion channels in the activation of anabolic signaling in skeletal muscle. Am J Physiol Endocrinol Metab. 2019 Jan 1;316(1):E86-E95. doi: 10.1152/ajpendo.00261.2018. Epub 2018 Nov 20. PubMed PMID: 30457911. View Article
- Hafner AS, Donlin-Asp PG, Leitch B, Herzog E, Schuman EM. Local protein synthesis is a ubiquitous feature of neuronal pre- and postsynaptic compartments. Science. 2019 May 17;364(6441). View Article
- Heumuller M, Glock C, Rangaraju V, Biever A , Schuman EM. A genetically encoadable cell-type-specific protein synthesis inhibitor. Nat Methods. 2019. View Article
- Kim JK, Cho J, Kim SH, et al. Brain somatic mutations in MTOR reveal translational dysregulations underlying intractable focal epilepsy. J Clin Invest. 2019;129(10):4207-4223. View Article
- Ding Q, Sethna F, Wu XT, et al. Transcriptome signature analysis repurposes trifluoperazine for the treatment of fragile X syndrome in mouse model. Commun Biol. 2020;3(1):127. Published 2020 Mar 16. View article
- Weiterer SS, Meier-Soelch J, Georgomanolis T, et al. Distinct IL-1α-responsive enhancers promote acute and coordinated changes in chromatin topology in a hierarchical manner. EMBO J. 2020;39(1):e101533. View article
- Crowell KT, Moreno S, Steiner JL, Coleman CS, Soybel DI, Lang CH. Temporally Distinct Regulation of Pathways Contributing to Cardiac Proteostasis During the Acute and Recovery Phases of Sepsis. Shock. 2018;50(6):616-626. View article
- Kitakaze K, Taniuchi S, Kawano E, et al. Cell-based HTS identifies a chemical chaperone for preventing ER protein aggregation and proteotoxicity. Elife. 2019;8:e43302. Published 2019 Dec 17. View article
- Shah DS, Nisr RB, Stretton C, Krasteva-Christ G, Hundal HS. Caveolin-3 deficiency associated with the dystrophy P104L mutation impairs skeletal muscle mitochondrial form and function. J Cachexia Sarcopenia Muscle. 2020;11(3):838-858. View article
- Ding Q, Sethna F, Wu XT, et al. Transcriptome signature analysis repurposes trifluoperazine for the treatment of fragile X syndrome in mouse model. Commun Biol. 2020;3(1):127. Published 2020 Mar 16. View article
- Cui D, Drake JC, Wilson RJ, et al. A novel voluntary weightlifting model in mice promotes muscle adaptation and insulin sensitivity with simultaneous enhancement of autophagy and mTOR pathway. FASEB J. 2020;34(6):7330-7344. View article
- Rodrigues DC, Harvey EM, Suraj R, et al. Methylglyoxal couples metabolic and translational control of Notch signalling in mammalian neural stem cells. Nat Commun. 2020;11(1):2018. Published 2020 Apr 24. View article
- Raimer AC, Singh SS, Edula MR, et al. Temperature-sensitive spinal muscular atrophy-causing point mutations lead to SMN instability, locomotor defects and premature lethality in Drosophila. Dis Model Mech. 2020;13(5):dmm043307. Published 2020 May 22. View article
- Ghosh A, Mizuno K, Tiwari SS, et al. Alzheimer's disease-related dysregulation of mRNA translation causes key pathological features with ageing. Transl Psychiatry. 2020;10(1):192. Published 2020 Jun 16. View article
- Ho JJD, Balukoff NC, Theodoridis PR, et al. A network of RNA-binding proteins controls translation efficiency to activate anaerobic metabolism. Nat Commun. 2020;11(1):2677. Published 2020 May 29. View article
- Ghosh A, Mizuno K, Tiwari SS, et al. Alzheimer's disease-related dysregulation of mRNA translation causes key pathological features with ageing. Transl Psychiatry. 2020;10(1):192. Published 2020 Jun 16. View article
- Rajgor D, Purkey AM, Sanderson JL, et al. Local miRNA-Dependent Translational Control of GABAAR Synthesis during Inhibitory Long-Term Potentiation. Cell Rep. 2020;31(12):107785. View article
- Goldsmith J, Marsh T, Asthana S, et al. Ribosome profiling reveals a functional role for autophagy in mRNA translational control. Commun Biol. 2020;3(1):388. Published 2020 Jul 17. View article
- Wuerth JD, Habjan M, Kainulainen M, et al. eIF2B as a Target for Viral Evasion of PKR-Mediated Translation Inhibition. mBio. 2020;11(4):e00976-20. Published 2020 Jul 14. View article
- Hörnberg H, Pérez-Garci E, Schreiner D, et al. Rescue of oxytocin response and social behaviour in a mouse model of autism. Nature. 2020;584(7820):252-256. View article
- Popova S, Ulanova A, Gritsyna Y, Salmov N, Rogachevsky V, Mikhailova G, Bobylev A, Bobyleva L, Yutskevich Y, Morenkov O, Zakharova N, Vikhlyantsev I. Predominant synthesis of giant myofibrillar proteins in striated muscles of the long-tailed ground squirrel Urocitellus undulatus during interbout arousal. Sci Rep. 2020 Sep 16;10(1):15185. View article
- Costa G, Bradbury JJ, Tarannum N, Herbert SP. RAB13 mRNA compartmentalisation spatially orients tissue morphogenesis. EMBO J. 2020 Sep 18:e106003. View article
- Popova S, Ulanova A, Gritsyna Y, et al. Predominant synthesis of giant myofibrillar proteins in striated muscles of the long-tailed ground squirrel Urocitellus undulatus during interbout arousal. Sci Rep. 2020;10:15185. Published 2020 Sep 16. View article
- Frolinger T, Smith C, Cobo CF, Sims S, Brathwaite J, de Boer S, Huang J, Pasinetti GM. Dietary polyphenols promote resilience against sleep deprivation-induced cognitive impairment by activating protein translation. FASEB J. 2018 Oct;32(10):5390-5404. View article
- Ballarò R, Beltrà M, De Lucia S, Pin F, Ranjbar K, Hulmi JJ, Costelli P, Penna F. Moderate exercise in mice improves cancer plus chemotherapy-induced muscle wasting and mitochondrial alterations. FASEB J. 2019 Apr;33(4):5482-5494. View article
- Middleton EA, Rowley JW, Campbell RA, Grissom CK, Brown SM, Beesley SJ, Schwertz H, Kosaka Y, Manne BK, Krauel K, Tolley ND, Eustes AS, Guo L, Paine R 3rd, Harris ES, Zimmerman GA, Weyrich AS, Rondina MT. Sepsis alters the transcriptional and translational landscape of human and murine platelets. Blood. 2019 Sep 19;134(12):911-923. View article
- Hu HJ, Deng XW, Li RX, Chen DW, Xue C. Inhibition of protein kinase C activity inhibits osteosarcoma metastasis. Arch Med Sci. 2019 Jul;15(4):1028-1034. View article
- Tyganov SA, Mochalova EP, Belova SP, Sharlo KA, Rozhkov SV, Vilchinskaya NA, Paramonova II, Mirzoev TM, Shenkman BS. Effects of Plantar Mechanical Stimulation on Anabolic and Catabolic Signaling in Rat Postural Muscle Under Short-Term Simulated Gravitational Unloading. Front Physiol. 2019 Sep 27;10:1252. View article
- Morrissey JA, Bigus E, Necarsulmer JC, Srinivasan V, Peppercorn K, O'Leary DJ, Mockett BG, Tate WP, Hughes SM, Parfitt KD, Abraham WC. The Tripeptide RER Mimics Secreted Amyloid Precursor Protein-Alpha in Upregulating LTP. Front Cell Neurosci. 2019 Oct 18;13:459. View article
- Farley-Barnes KI, Deniz E, Overton MM, Khokha MK, Baserga SJ. Paired Box 9 (PAX9), the RNA polymerase II transcription factor, regulates human ribosome biogenesis and craniofacial development. PLoS Genet. 2020 Aug 19;16(8):e1008967. View article
- Alvarez-Castelao B, Tom Dieck S, Fusco CM, Donlin-Asp P, Perez JD, Schuman EM. The switch-like expression of heme-regulated kinase 1 mediates neuronal proteostasis following proteasome inhibition. Elife. 2020 Apr 24;9:e52714. View article
- Balukoff NC, Ho JJD, Theodoridis PR, Wang M, Bokros M, Llanio LM, Krieger JR, Schatz JH, Lee S. A translational program that suppresses metabolism to shield the genome. Nat Commun. 2020 Nov 13;11(1):5755. View article
- Krause AR, Speacht TA, Steiner JL, Lang CH, Donahue HJ. Mechanical loading recovers bone but not muscle lost during unloading. NPJ Microgravity. 2020 Dec 3;6(1):36. View article
- Ding Q, Zhang F, Feng Y, Wang H. Carbamazepine Restores Neuronal Signaling, Protein Synthesis, and Cognitive Function in a Mouse Model of Fragile X Syndrome. Int J Mol Sci. 2020 Dec 7;21(23):9327. View article
- Tsai YL, Coady TH, Lu L, Zheng D, Alland I, Tian B, Shneider NA, Manley JL. ALS/FTD-associated protein FUS induces mitochondrial dysfunction by preferentially sequestering respiratory chain complex mRNAs. Genes Dev. 2020 Jun 1;34(11-12):785-805. View article
- Johnson B, VanBlargan LA, Xu W, White JP, Shan C, Shi PY, Zhang R, Adhikari J, Gross ML, Leung DW, Diamond MS, Amarasinghe GK. Human IFIT3 Modulates IFIT1 RNA Binding Specificity and Protein Stability. Immunity. 2018 Mar 20;48(3):487-499.e5. View article
- Donlin-Asp PG, Polisseni C, Klimek R, Heckel A, Schuman EM. Differential regulation of local mRNA dynamics and translation following long-term potentiation and depression. Proc Natl Acad Sci U S A. 2021 Mar 30;118(13):e2017578118. View article
If you publish research with this product, please let us know so we can cite your paper.

![Anti-Puromycin [3RH11] Antibody, 100ug Anti-GFP (Chicken-Standard) Antibody](https://www.kerafast.com/MediaStorage/Product/Images/Medium/113_200120200125541120D.jpg)

