Endoglycosidase F1 (Endo F1)
Endoglycosidase F1 (Endo F1) from Elizabethkingia miricola, is an enzyme used for removing N-linked oligosaccharides from glycoproteins.
Endoglycosidase F1, expressed in E. coli, cleaves asparagine linked or free high mannose (oligomannose) and hybrid structures. It differs from exoglycosidase, that is does not do so at the terminal residue. This enzyme is suitable for deglycoslylation of N-linked oligosaccharides from glycoproteins under native conditions, and is appropriate for a variety of glycomics, proteomics, and mass spectroscopy applications.
From the laboratory of Davide Comoletti, DVM, PhD, Rutgers University
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
Endoglycosidase F1 (Endo F1) from Elizabethkingia miricola, is an enzyme used for removing N-linked oligosaccharides from glycoproteins.
Endoglycosidase F1, expressed in E. coli, cleaves asparagine linked or free high mannose (oligomannose) and hybrid structures. It differs from exoglycosidase, that is does not do so at the terminal residue. This enzyme is suitable for deglycoslylation of N-linked oligosaccharides from glycoproteins under native conditions, and is appropriate for a variety of glycomics, proteomics, and mass spectroscopy applications.
From the laboratory of Davide Comoletti, DVM, PhD, Rutgers University
 Part of The Investigator's Annexe program.
Part of The Investigator's Annexe program.
| Product Type: | Protein |
| Name: | Endoglycosidase F1 (endo F) |
| Accession ID: | P36911 |
| Source: | Elizabethkingia miricola |
| Molecular Weight: | 63 kDa (with the Tag) |
| Fusion Tag(s): | GST |
| Buffer: | 20 mM TRIS pH 8.0, 150 mM NaCl, 1 mM DTT, 20 % Glycerol |
| Concentration: | 8mg/mL |
| Comments: | Plasmid pGEX 3X, Expressed in E. coli BL21 (DE3) |
| Storage: | -80C |
| Shipped: | Dry ice |
Protocol Notes
For efficient deglycosylation, use overnight incubation (~16 h) at 4 deg C with a ratio 1:20 (ug enzyme:ug target protein), in mid pH conditions (Suggested; 50 mM TRIS pH 7.5, 150 mM NaCl).
SDS PAGE
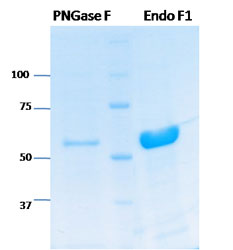
SDS-PAGE analysis showing purification of PNGase F on the left, Ladder in the middle, and Endo F1 on the right.
- Ao J, Chinnici JL, Maddi A, Free SJ. The N-Linked Outer Chain Mannans and the Dfg5p and Dcw1p Endo-α-1,6-Mannanases Are Needed for Incorporation of Candida albicans Glycoproteins into the Cell Wall. Eukaryot Cell. 2015 Aug;14(8):792-803. View Article
If you publish research with this product, please let us know so we can cite your paper.